Telfa® AMD™ Antimicrobial Dressings
Telfa® AMD™ Antimicrobial Dressings are impregnated with polyhexamethylene biguanide (PHMB) to provide an antimicrobial barrier to prevent microbial penetration and microbial growth within the dressing. Product is gentle to normal skin flora.

Cardinal Health
Cardinal Health is focused on improving the cost-effectiveness of health care through a suite of advanced solutions designed to improve supply chain efficiency, optimize business processes and performance, deliver value-oriented medical products and pharmaceuticals and connect clinicians and patients for streamlined care and better wellness.Benefits
• Perforated mylar film with absorbent core allows it to be used as primary dressing
• Contains PHMB (polyhexamethylene biguanide), a gentle but effective antiseptic
• PHMB destroys the cell wall, making resistance very unlikely
• Limits cross-contamination and reduces risk of infection
• Non-adherent for less painful dressing changes
• Resists bacterial penetration and colonization
• Highly effective against most organisms
Indications
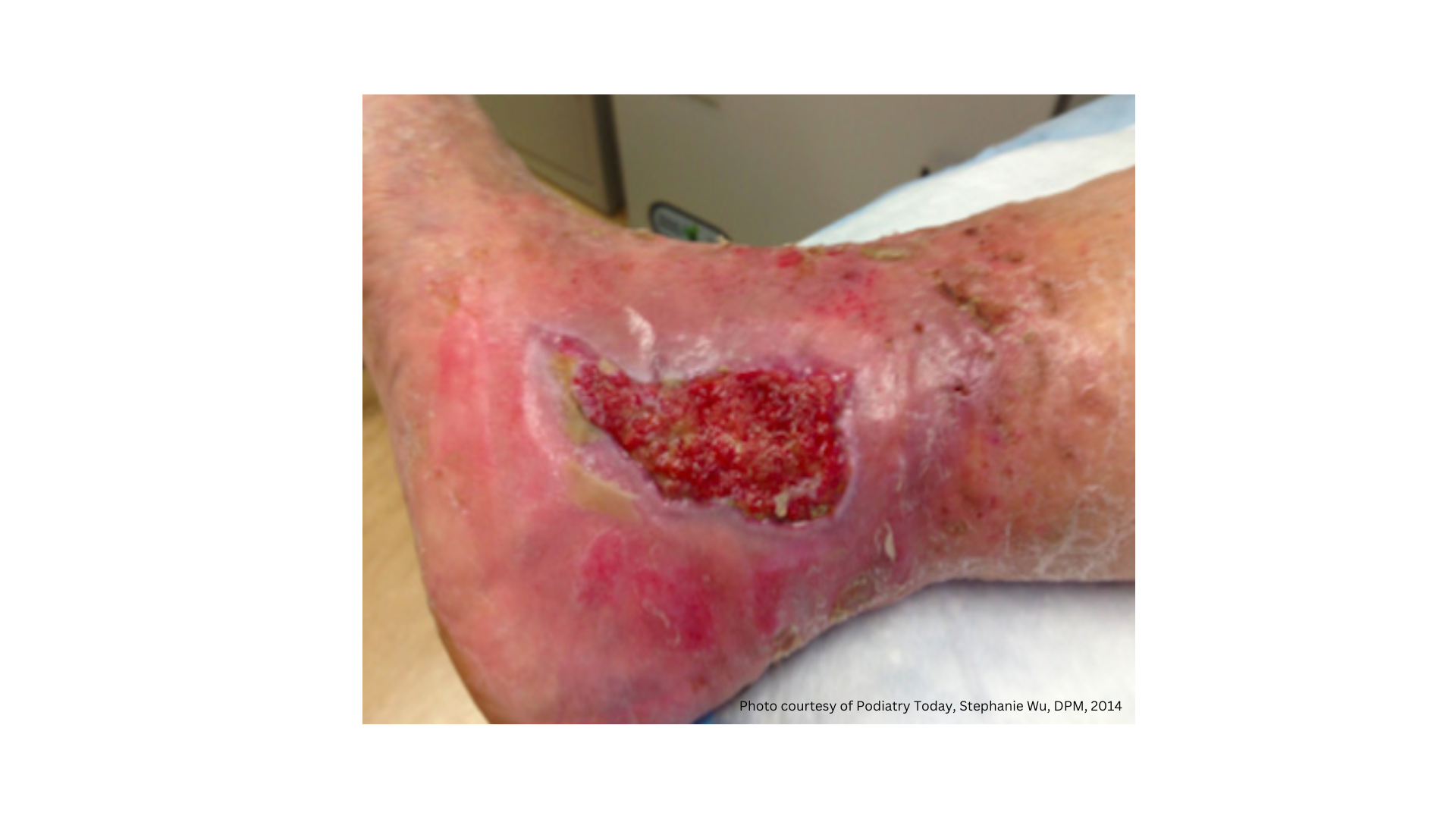
Telfa® AMD™ Antimicrobial Dressings can be used as a primary dressing to minimize bacterial log counts in the wound bed, or as a secondary dressing to eliminate growth within the gauze and protect against bacterial proliferation through the gauze.
Warnings and Precautions
Dakin's Solution cleanser may inactivate PHMB. This will impact dressing efficacy but should not be dangerous to patient health.
How Supplied/Sizing
Product features
Other features
Recommended Use
Infected Wounds
Non/Minimally Exuding Wounds
Superficial Wounds
Mode of Use/Application
No change to clinical protocol - can be used for all applications to a standard Telfa™ dressing.
No posters match your selected filters. Remove some filtres, or reset them and start again.
Have a product to submit?
Be included in the most comprehensive wound care products directory
and online database.
Learn More