ZeniFOAM-Ag GENTLE BORDER
ZeniFOAM-Ag GENTLE BORDER is a sterile, silicone-faced, foam dressing with a silicone border used for moderate to heavy wound drainage. Multilayered to provide maximum absorption, protection and up to 7 days antibacterial effectiveness.
Focus Health Group
ZeniMedical is a privately owned and operated company with a product line of wound treatment products.
Benefits
• Multilayered high performance dressing
• Highly absorbent
• Protects against maceration
• Gentle silicone adhesive
• Non-adherent
• Island dressing design for easy application
• Broad-spectrum antimicrobial
• Can be repositioned after application
• Highly conformable
• Waterproof backing
• Antimicrobial effectiveness for up to 7 days
Indications
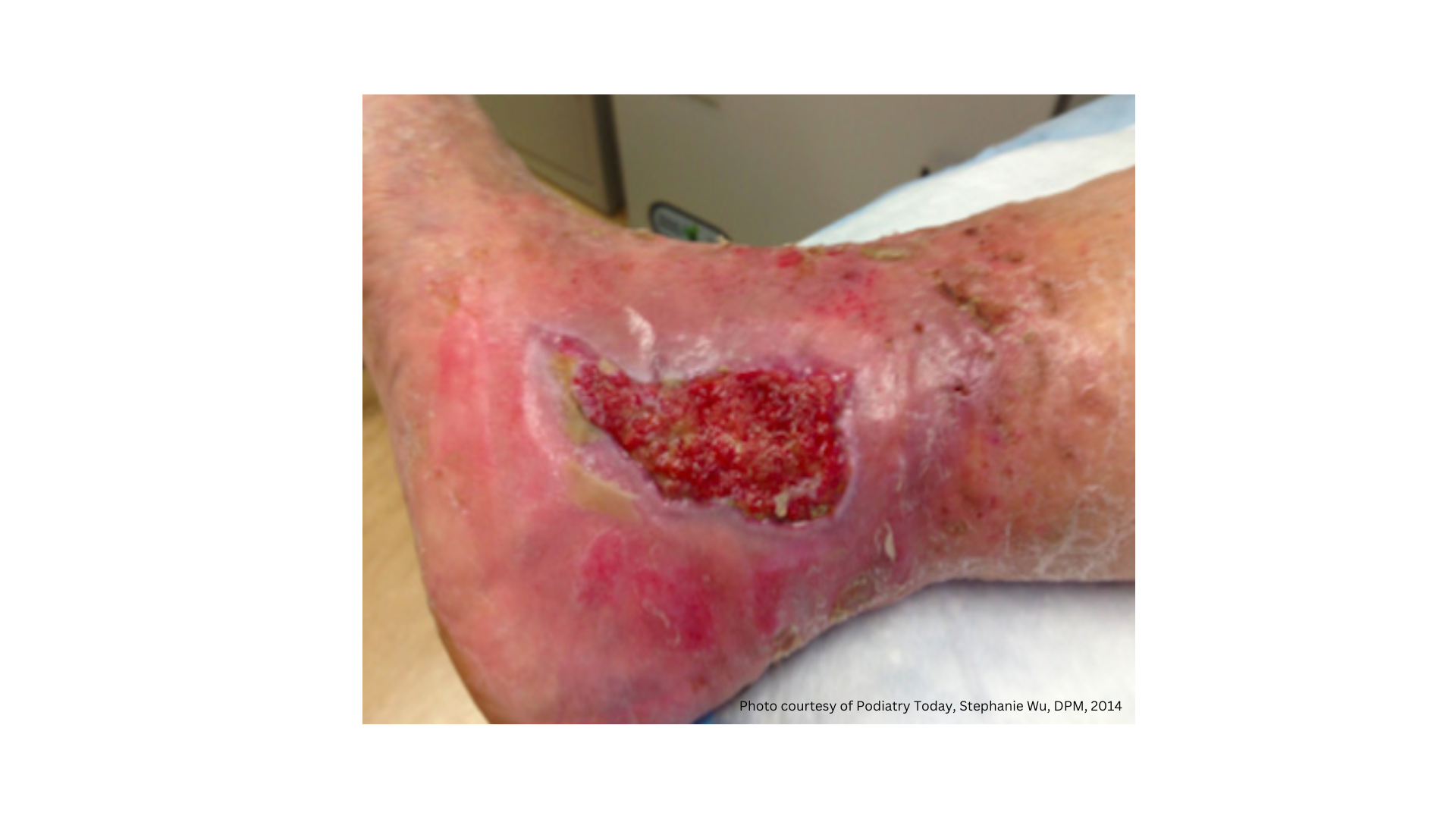
ZeniFOAM-Ag GENTLE BORDER is for use on moderate to heavy draining wounds, pressure injuries, superficial burns, leg ulcers, traumatic wounds, partial-thickness burns, foot ulcers, and surgical wounds.
Contraindications
Do not use as an internal dressing/sponge.
Do not use on third-degree burns.
Do not use if a known allergy or hypersensitivity exists silver.
Warnings and Precautions
ZeniFOAM-Ag GENTLE BORDER requires a prescription for use from a medical provider.
ZeniFOAM-Ag GENTLE BORDER is single use only. Do not reuse.
Do not use if the pouch is damaged or opened.
Not recommended for use on dry wounds.
Do not use ZeniFOAM-Ag GENTLE BORDER with oxidizing agents such as hypochlorite solutions or hydrogen peroxide.
ZeniFOAM-Ag GENTLE BORDER may cause transient discoloration of the wound bed and surrounding skin. Frequent or prolonged use may result in permanent discoloration of the skin.
ZeniFOAM-Ag GENTLE BORDER does not replace the need for systemic therapy or other adequate infection treatment in the event of clinical infection.
Do not use during radiation treatment or examinations such as x-ray, magnetic resonance imaging (MRI) or ultrasound.
Avoid contact with EKG electrodes or conductive gels during electronic measurements such as electrocardiograms (ECG) and electroencephalogram (EEG).
ZeniFOAM-Ag GENTLE BORDER is intended for short term use up to 28 days.
Storage Requirements
Store ZeniFOAM-Ag GENTLE BORDER in a cool, dry place and avoid light.
Active Ingredients
Ionic silver
How Supplied/Sizing
Product features
Other features
Recommended Use
Acute Wounds
Burns
Chronic Wounds
Dehisced Wounds
Diabetic Foot
Granulating/Epithelializing Wounds
Infected Wounds
Moderate/Highly Exudating Wounds
Palliative Wounds
Pressure Ulcers
Sloughy Wounds
Superficial Wounds
Surgical Wounds
Venous Ulcers
Mode of Use/Application
The sticky side of the dressing is the silicone contact layer and it should be placed touching the wound. Please note: Silicone is naturally sticky to the touch; however, when in contact with wound drainage, the silicone is non-adherent to the wound. Unlike traditional glue adhesives, ZeniFOAM Ag GENTLE BORDER is gentle and easy to remove from the skin around the wound. The colored backing on ZeniFOAM Ag GENTLE BORDER is waterproof to prevent drainage from striking through the dressing and to provide protection from external contaminants.
Removal & Change Frequency
Replace dressing every other day, when drainage is visible to the edge of the foam pad, if the dressing leaks, or as instructed by your medical provider.
Construction
Silicone-faced contact layer, absorbent polyurethane foam with ionic silver, non-woven layer, super absorbent core, waterproof, breathable film backing and border
Clinically Tested
Latex-friendly
Non-allergenic
Non-cytotoxic
Non-irritating
No posters match your selected filters. Remove some filtres, or reset them and start again.
Have a product to submit?
Be included in the most comprehensive wound care products directory
and online database.
Learn More