Cellesta™ Amniotic Membrane
Cellesta™ Amniotic Membrane is a minimally manipulated, placental allograft product. This natural human tissue scaffold with relevant, inherent characteristics can be utilized for therapeutic solutions.
Ventris Medical
Ventris Medical is a Newport Beach-based health care company, currently offering its full line of Cellesta™ branded placental allograft products.Benefits
• Biological barrier with relevant, inherent characteristics often used for therepeutic solutions
• Natural human tissue scaffold shields and cushions, as well as acts as a matrix for selective cellular migration, just as it does in utero
• Available for a broad range of surgical applications, including general surgery, wound care, plastic surgery, and urologic and orthopedic procedures
Indications
Cellesta™ Amniotic Membrane functions as a covering or barrier that offers protection from the surrounding environment in reparative and reconstructive procedures.
Storage Requirements
Store at ambient temperature.
How Supplied/Sizing
Product features
Recommended Use
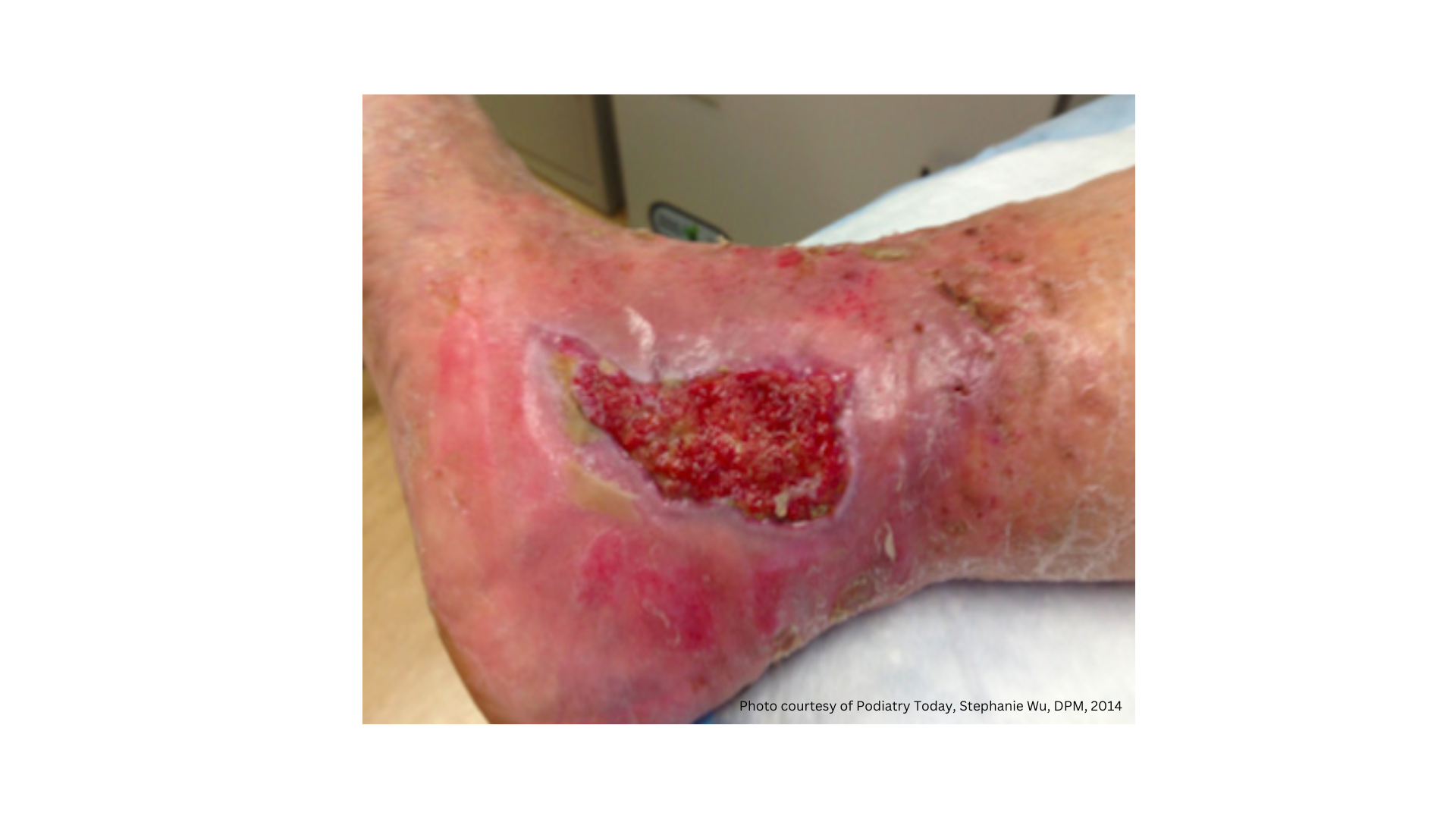
Acute Wounds
Cavity Wounds
Chronic Wounds
Deep Wounds
Diabetic Foot
Pressure Ulcers
Surgical Wounds
Venous Ulcers
Mode of Use/Application
Membrane may be sutured, glued, or laid over desired tissue. Once the poly mesh backing is removed, either side of the graft may be applied to the target tissue, as the grafts are provided orientation-neutral.
No posters match your selected filters. Remove some filtres, or reset them and start again.
Have a product to submit?
Be included in the most comprehensive wound care products directory
and online database.
Learn More